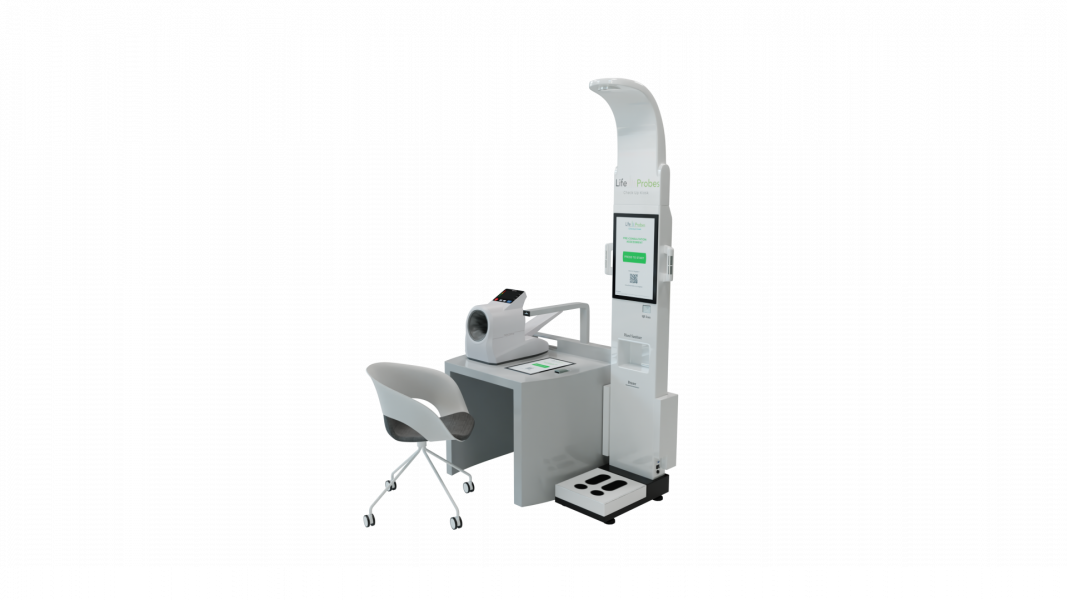
Northwestern Medicine, a nationally ranked hospital in Chicago, Illinois, has deployed LifeProbes kiosks in its facilities. The kiosks were piloted in the Northwestern Innovation Center for 6 months and are now fully integrated with the Epic electronic medical record (EMR). The kiosks use automated technology to collect patient vital signs, which saves time for healthcare […]
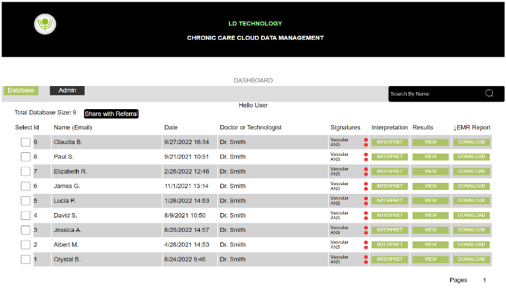
We are excited to announce the release of TM-Flow version 7, which has been two years in the making. This new version integrates the CCS Cloud, providing users with a number of improvements, including: The ability to read and print exams from any tablet or smartphone, securely stored in the cloud. The ability to make […]
Tuesday, May 26, 2020
LD Technology is pleased to announce the release of the Lifeprobes Kiosk following two new 510k clearances ( K200141 and K 200287) .
The Lifeprobes Kiosk performs automated pre-consultation patient assessments that allow healthcare professionals to maximize operational efficiencies by improving the speed and flow rate of patient onboarding in medical offices, clinics, and hospitals
n order to help our customers in this difficult time , we supply new high quality products at prefential price:
Surgical Masks, RTD serological antibody kits and UV C Sterilizer. I.e. COVID-19 products Contact us for prices
Near Infrared Therapy is an innovative pain management device for small fiber neuropathy symptoms. The Neuro Pain Care product combines the SweatC and the Near Infrared Therapy device to evaluate the improvement of the microcirculation and pain relief.
LD Technology, LLC. is pleased to announce the successful issue of the
Patent No.: US 10,383,565 B2.
In an ongoing effort to provide continuous improvements to our quality system and products, while also responding to our customer’s feedback, we are proud to release the latest version of our TM-Flow System - Version 5.
We want to thank you for attending the LD TECHNOLOGY Symposium on October 18-19th 2018.
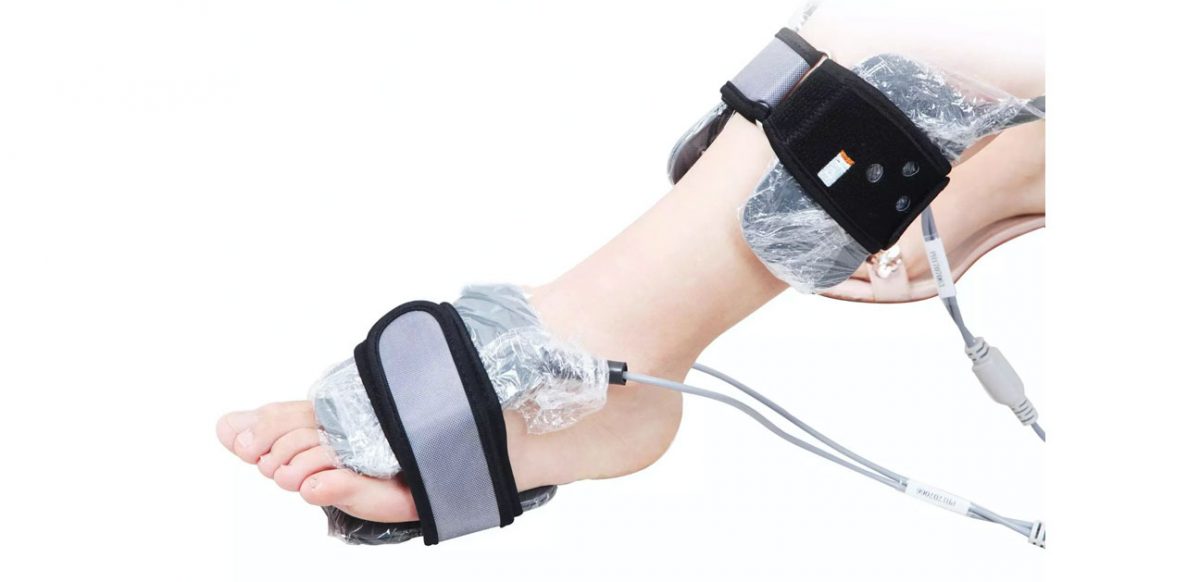
The new TM-Flow wireless increases the patient comfort and reduces the time and placement of the cuffs for the technician before the measurement